The best way to prevent hydrate formation (and corrosion) is to keep pipelines, tubing and equipment dry of liquid water. There are occasions, right or wrong, when the decision is made to operate a line or process containing liquid water. If this decision is made, and the process temperature is below the hydrate point, inhibition of this water is necessary. This is of particular interest in gas gathering systems [1] and subsea operations [2] during normal production as well as during shut down.
Many materials may be added to water to depress both the hydrate and freezing temperatures. For many practical reasons, a thermodynamic hydrate inhibitor (THI) such as an alcohol or one of the glycols is injected, usually methanol, diethylene glycol (DEG) or monoethylene glycol (MEG). All may be recovered and recirculated, but the economics of methanol recovery may not be favorable in many cases. Hydrate prevention with methanol and or glycols can be quite expensive because of the high effective dosage required (10% to 60% of the water phase). Large concentrations of solvents aggravate potential scale problems by lowering the solubility of scaling salts in water and precipitating most known scale inhibitors. The total injection rate of inhibitor required is the amount/concentration of inhibitor in the liquid water phase for the desired hydrate temperature suppression, plus the amount of inhibitor that will distribute in the vapor and liquid hydrocarbon phases. Any inhibitor in the vapor phase or liquid hydrocarbon phase has little effect on hydrate formation conditions. Due to the accuracy limitations of the hydrate depression calculations and flow distribution in the process, it is recommended that the hydrate formation temperature with inhibition be chosen with a design factor below the coldest expected operating temperature of the system to ensure adequate inhibitor injection rates.
Solubility loss of MEG in the gas phase is negligible and loss to the liquid hydrocarbon phase is very low, 3.5 L/106Sm3 (0.23 lbm/MMscf) [3]. Methanol losses are more significant, particularly vapor phase losses. Based on Figure 6.20 in reference [3], depending on operating conditions, the solubility loss of methanol into the gas phase can be very high, typically 16 mg/Sm3 (1 lbm/MMscf) for every percent methanol in water phase. Losses to the liquid hydrocarbon are higher than for MEG but usually less than 1-2 % of hydrocarbon volume. At typical pipeline inhibition conditions, a solubility of about 0.4 kg/m3 (0.15 lbm/bbl) is generally adequate for planning purposes [3]. Depending on solubility losses, chemical makeup requirements for methanol can be very large and expensive for both once-through systems and methanol recovery units. In addition, the downstream processes like petrochemical and LNG plants cannot tolerate methanol in the feed gas.
Determination of the amount and concentration of inhibitors and their distribution in different phases is very important for practical purposes and industrial applications. Therefore, to determine the required amount and concentration of these inhibitors, several thermodynamic models for hand and rigorous calculations have been developed and incorporated into computer software [4].
As previously stated, a significant amount of methanol would be lost to the hydrocarbon phases, which may cause problems for refineries, petrochemical, and gas plants downstream. In gas plants where there is propane recovery the methanol will follow the propane product and be a potential cause for propane to go off specification. Methanol has also been known to cause premature failure in molecular sieves. In refineries the methanol must be washed out of the crude/condensate, where it presents a problem in wastewater treatment. In petrochemical plants methanol is also considered poison for catalysts.
In offshore production, gas lift and gas injection for pressure maintenance are becoming common practice. The associated gas produced with crude oil and water is separated, compressed and normally dehydrated with triethylene glycol (TEG) before injection or export. Since the produced oil/water/gas lines pose hydrate formation during normal production or shut downs, methanol is commonly injected to prevent hydrate formation and plugging of flow lines and gas lift/reinjection lines. Occasionally, TEG dehydration units shut down or may produce off spec dry gas which requires methanol injection. Consequently, some of the injected methanol ends up back in the produced oil/water/gas stream.
In this TOTM we will consider the presence of methanol in the produced oil/water/gas stream and determine the quantitative traces of methanol ending up in the TEG dehydrated gas. To achieve this, we simulated by computer an offshore production facility consisting of oil/water/gas multistage-separation, compression and TEG dehydration processes and determined the methanol concentration in the dried gas. We also studied the effect of wet gas temperature, the number of theoretical trays in the TEG contactor, the water content spec of dry gas, and lean TEG circulation rate on the dried gas methanol content. For this purpose methanol content in the production stream was assumed to vary from zero to 350 PPM (V).
Case Study:
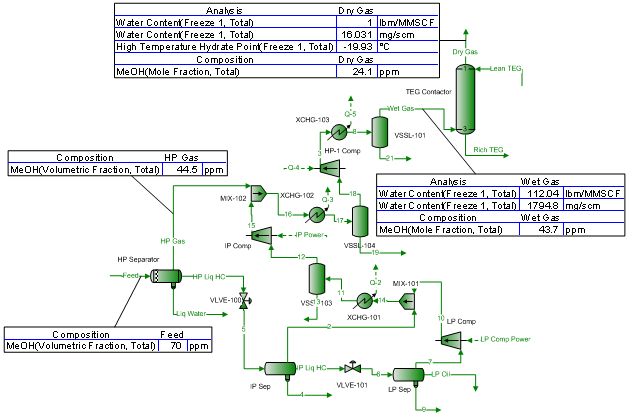
A simplified process flow diagram (PFD) for the offshore production facilities considered in this study is shown in Figure 1. The production stream (oil, water, gas, and methanol) was passed through the high pressure separator where free water and gas were separated and the oil was passed through the intermediate and low pressure separators for subsequent gas separation from oil. The separators’ off gas streams were recompressed and cooled to 48 bara and 35 °C (696 psia and 95 °F) before entering the TEG contactor for dehydration. The dried gas was compressed further (not shown in the PFD) to 232 bara (3365 psia) for reinjection or export purposes. To meet a water content spec of 32 mg/Sm3 (2 lbm/MMscf) or lower, a lean TEG concentration of 99.95 weight percent was used in all of the simulation runs.
To study the impact of methanol (MeOH) concentration and determine its traces in the TEG dehydrated gas, the MeOH content of the production stream feed to the high pressure separator was assumed to vary from 0 to 350 PPM (V). This variation of MeOH content was chosen due to the uncertainty of its concentration in the production stream. The wet compressed gas temperature is an important parameter in the operation of a TEG unit and affects the water content of dried gas and the required lean TEG solution circulation rate and/or the number of required theoretical trays. Depending on the design and/or operational problem like scaling on the cooling side of the gas cooler, the wet gas temperature may be higher than 35 °C (95 °F). Therefore, the wet gas temperature was assumed to vary from 35 to 50 °C with 5 °C increment (95 to 122 °F and 9 °F increment). Depending on the requirement, 2 or 3 trays theoretical was used in the contactor unit. For each case the lean TEG solution rate was varied to meet the desired water content specification for each case.
Figure 1. Simple process flow diagram used in this case study
Results and Discussion:
The ProMax simulation software [6] was used to perform computer simulations for different cases of interest and determined the concentration/traces of MeOH in the TEG dehydrated gas. Twelve cases were studied in which the number of theoretical trays, wet feed gas temperature and pressure, dried gas water content, lean TEG solution weight percent, circulation rate, pressure and temperature were specified. For each case the MeOH content in the feed to the high pressure (HP) separator was assumed to vary from 0 to 350 part per million by volume, PPM (V) and the corresponding MeOH concentration in dried gas was determined. Table 1 presents the computer simulation results for one the 12 case studies. The absorption % presented in the last two columns of Table 1 is defined as:
Overall % = 100 (MeOH PPM in feed to the HP Separator – MeOH PPM in dry gas) /( MeOH PPM in feed to HP Separator)
TEG Contactor % = 100 (MeOH PPM in wet gas – MeOH PPM in dry gas) /( MeOH PPM in wet gas).
Notice the calculated MeOH absorption percents are relatively constant and independent of MeOH PPM in the feed to HP separator or wet gas. As shown in the last row of Table 1, the process overall and TEG contactor MeOH absorption percents are 61.1, and 30.6, respectively.
Table 1. Typical computer simulation results
| Lean TEG Wt%=99.95 | Lean TEG Solution Std Liquid Vol Rate = 2.91 m3/h (16.9 L TEG/kg Water) | ||||||||
| No. of Theoretical Tray=3 | Lean TEG Solution Temperature = 38 °C | ||||||||
| Wet Gas To TEG T, °C | MeOH, PPM (V) | Dry Gas | MeOH Absorption | ||||||
| In the Feed of Inlet HP Separator | In Wet Gas To TEG | In TEG Dry Gas | Hydrate Formation T, °C | Water Content lbm/MMSCF | Water Content mg/Sm3 | TEG Contactor % | Overall % | ||
| 35 | 0 | 0 | 0.0 | -20.0 | 1.00 | 16.0 | NA | NA | |
| 25.6 | 14.4 | 10.0 | -20.0 | 1.00 | 16.0 | 30.6 | 60.9 | ||
| 70.0 | 39.4 | 27.4 | -20.0 | 1.00 | 15.9 | 30.5 | 60.8 | ||
| 104.9 | 59.0 | 41.0 | -20.0 | 0.99 | 15.9 | 30.5 | 60.9 | ||
| 139.8 | 78.5 | 54.5 | -20.0 | 0.99 | 15.9 | 30.6 | 61.0 | ||
| 174.6 | 97.9 | 68.0 | -20.1 | 0.99 | 15.8 | 30.5 | 61.1 | ||
| 209.4 | 117.0 | 81.4 | -20.1 | 0.99 | 15.8 | 30.4 | 61.1 | ||
| 244.2 | 137.0 | 94.8 | -20.1 | 0.98 | 15.8 | 30.8 | 61.2 | ||
| 278.9 | 156.0 | 108.0 | -20.1 | 0.98 | 15.7 | 30.8 | 61.3 | ||
| 313.6 | 175.0 | 121.0 | -20.1 | 0.98 | 15.7 | 30.9 | 61.4 | ||
| 348.3 | 194.0 | 134.0 | -20.2 | 0.98 | 15.7 | 30.9 | 61.5 | ||
| Average | 30.6 | 61.1 | |||||||
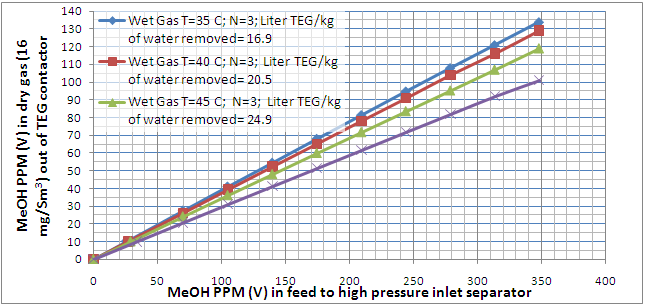
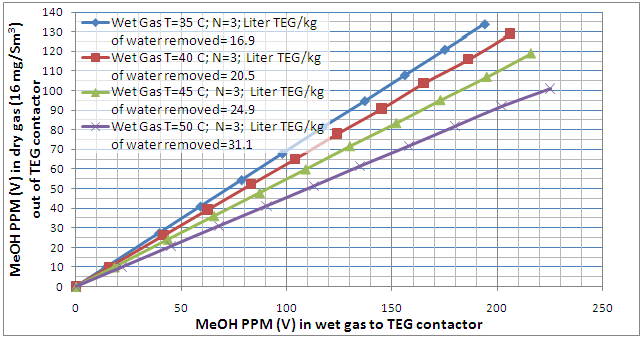
The MeOH concentration profile in the dried gas as a function of MeOH concentration in the feed to HP separator and MeOH concentration in the wet gas for 4 of these cases are presented in Figures 2 and 3, respectively. Notice the number of theoretical trays (N=3) and dried gas water content spec (16 mg/Sm3) were kept constant in these two figures. These two figures indicate that as the wet gas temperature increases, the lean TEG solution increases; therefore, more MeOH is picked up (absorbed) by TEG solution. This is explained by the fact that as the wet gas temperature increases, its capacity to hold water vapor increases. Since the dry gas water content spec had to remain constant, more lean TEG solution is required to remove the water. Because of higher lean TEG circulation rate, more MeOH is also absorbed in the contactor.
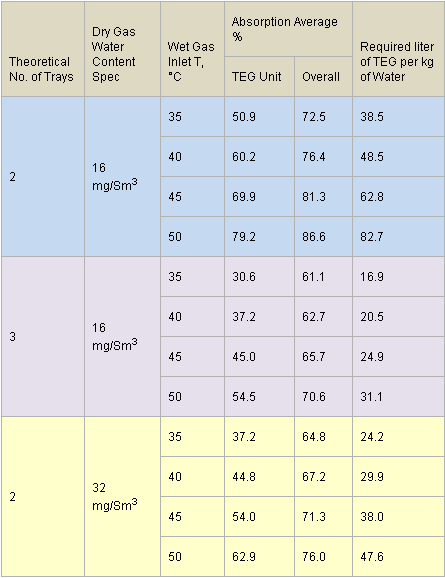
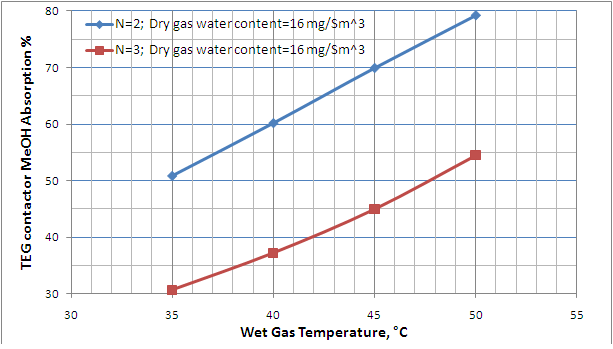
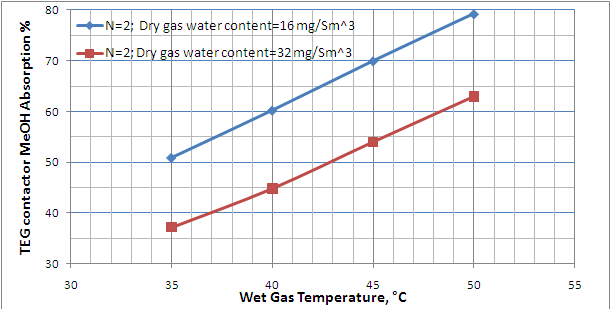
Table 2 presents the summary results for all 12 cases investigated. As shown in this table, for dry gas water content spec of 16 mg/Sm3 (1 lbm/MMscf), in comparison to three theoretical trays, the case of two theoretical trays requires a higher circulation rate; therefore, the MeOH absorption factor increases drastically (lower traces of MeOH in dry gas). The MeOH absorption is a function of the TEG circulation rate through the contactor. The greater the circulation rate, the greater the absorption. This high MeOH absorption % is favorable for the downstream process units; however, for the cases of N=2 and wet gas temperatures of 45 and 50 °C, the required liter of TEG per kg of water removed is much higher than the recommended value. According to Chapter 18 of reference [5], the recommended range is from 16 to 50 liter of TEG per kg of water removed (2 to 6 gal TEG/lb water removed). For dry gas water content spec of 32 mg/Sm3(32 lbm/MMscf) and two theoretical trays, the required lean TEG circulation rate drops down within the recommended range for all four wet gas temperatures.
Figure 2. Variation of MeOH content of the TEG dehydrated gas as a function of MeOH content in the inlet separatoor feed and wet gas temperature
Figure 3. Variation of MeOH content of the TEG dehydrated gas as a function of the wet gas MeOH content temperature
Table 2. Summary of all simulation results
The results in Table 2 are also represented in Figures 4 and 5. These figures can be used to estimate quickly the MeOH content of TEG dehydrated gas for a specified number of theoretical trays, and the wet gas temperature and MeOH content.
In summary, a case study based on an offshore production facility was undertaken to investigate the impact of design and operational parameters on the trace of MeOH in the TEG dehydrated gas. The results can be summarized as follows:
- The MeOH concentration in TEG dehydrated gas is proportional to the MeOH concentration in the feed to HP separator or in the wet gas to the TEG contactor.
- The MeOH concentration in TEG dehydrated gas decreases as the wet gas temperature increases.
- The MeOH concentration in TEG dehydrated gas decreases as the theoretical number of trays decreases, (or as the TEG circulation rate increases).
Notice, the above results were drawn based on twelve simulation runs for a single case study. They may be used for general guidelines. We believe each specific case should be analyzed separately and thoroughly.
To learn more about similar cases and how to minimize operational problems, we suggest attending the John M. Campbell courses; G4 (Gas Conditioning and Processing) and G5 (Gas Conditioning and Processing-Special).
John M. Campbell Consulting (JMCC) offers consulting expertise on this subject and many others. For more information about the services JMCC provides, visit our website at www.jmcampbellconsulting.
By: Dr. Mahmood Moshfeghian
Reference:
- Bullin, K.A., Bullin, J.A., “Optimizing methanol usage for hydrate inhibition in a gas gathering system,” Presented at the 83rd Annual GPA Convention – March 15, 2004.
- Szymczak, S., Sanders, K., Pakulski, M., Higgins, T.; “Chemical Compromise: A Thermodynamic and Low-Dose Hydrate-Inhibitor Solution for Hydrate Control in the Gulf of Mexico,” SPE Projects, Facilities & Construction, (Dec 2006).
- Campbell, J. M., “Gas Conditioning and Processing”, Vol. 1, The Basic Principles, 8th Ed., Second Printing, J. M. Campbell and Company, Norman, Oklahoma, (2002).
- Asadi Zeydabadi, B., Haghshenas, M., Roshani, S., and Moshfeghian, M., “Prevent system hydrate formation during sudden depressurization,” Hydrocarbon Processing, J., pp 83-91, April 2006.
- Campbell, J. M., “Gas Conditioning and Processing”, Vol. 2, The Equipment Module, 8th Ed., Second Printing, J. M. Campbell and Company, Norman, Oklahoma, (2002).
- ProMax 3.1, Bryan Research and Engineering, Inc, Bryan, Texas, 2009.
Figure 4. Effect of wet gas temperature and number of theoretical trays absorption % on the TEG contactor MeOH absorption %
Figure 5. Effect of wet gas temperature and dry gas water content spec on the TEG contactor MeOH absorption %

Dear Sir,
Very interesting your papers regarding gas processing. We are working on a TEG dehydration unit for NG and CO2 and I would like to ask you if the presence of traces of methanol could have a negative influence on the TEG drying and regeneration process and if so which could be the limiting methanol concentration. Thank you very much.
Hey there! I’ve been following your web site for a long time now and finally got the bravery to go ahead and give you a shout out from Houston Texas! Just wanted to tell you keep up the fantastic work!
A lot of the things you say happens to be astonishingly precise and that makes me ponder the reason why I had not looked at this with this light previously. Your article truly did turn the light on for me personally as far as this specific subject goes. However at this time there is actually 1 factor I am not really too comfortable with so while I attempt to reconcile that with the central theme of your position, permit me see what the rest of the subscribers have to point out.Well done.
[…] October 2010 tip of the month (TOTM) considered the presence of methanol in the produced oil/water/gas stream and determined the […]
[…] M., October 2010 tip of the month, PetroSkills – John M. Campbell, […]
[…] M., October 2010 tip of the month, PetroSkills | John M. Campbell, […]
[…] M., October 2010 tip of the month, PetroSkills | John M. Campbell, […]
Hello Sir, I am in Cameroon, I am in internship in GAZ DU CAMEROUN and I am working on Hydrate formation, Gaz du Cameroun have a problem of hydrates formation; it injected methanol but the results in not concluant, Sir I want to know how make to prevent this hydrate. It is condensat gas reservoir.